
The scale has been named the Pauling scale in his honour. The most commonly used scale to measure electronegativity was designed by Linus Pauling. It is a dimensionless property because it is only a tendency. The tendency of an atom in a molecule to attract the shared pair of electrons towards itself is known as electronegativity. Main article: Electronegativity Periodic variation of Pauling electronegativities In that case, the ionization energy decreases as atomic size increases due to adding a valence shell, thereby diminishing the nucleus's attraction to electrons. However, suppose one moves down in a group. The decrease in the atomic size results in a more potent force of attraction between the electrons and the nucleus. Trend-wise, as one moves from left to right across a period in the modern periodic table, the ionization energy increases as the nuclear charge increases and the atomic size decreases. The energy needed to remove the second electron from the neutral atom is called the second ionization energy and so on. The first ionization energy is the amount of energy that is required to remove the first electron from a neutral atom. It is also referred to as ionization potential. 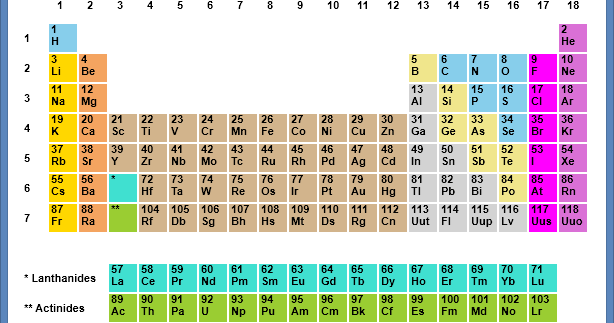
The ionization energy is the minimum amount of energy that an electron in a gaseous atom or ion has to absorb to come out of the influence of attracting force of the nucleus. These trends give a qualitative assessment of the properties of each element. These trends exist because of the similar electron configurations of the elements within their respective groups or periods they reflect the periodic nature of the elements. Major periodic trends include atomic radius, ionization energy, electron affinity, electronegativity, valency and metallic character. They were discovered by the Russian chemist Dmitri Mendeleev in the year 1863. In chemistry, periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of certain elements when grouped by period and/or group. Unfortunately, there was a slightly different system in place in Europe.Specific recurring patterns that are present in the modern periodic table The periodic trends in properties of elements. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. A new period begins when a new principal energy level begins filling with electrons. There are seven periods in the periodic table, with each one beginning at the far left. \) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
